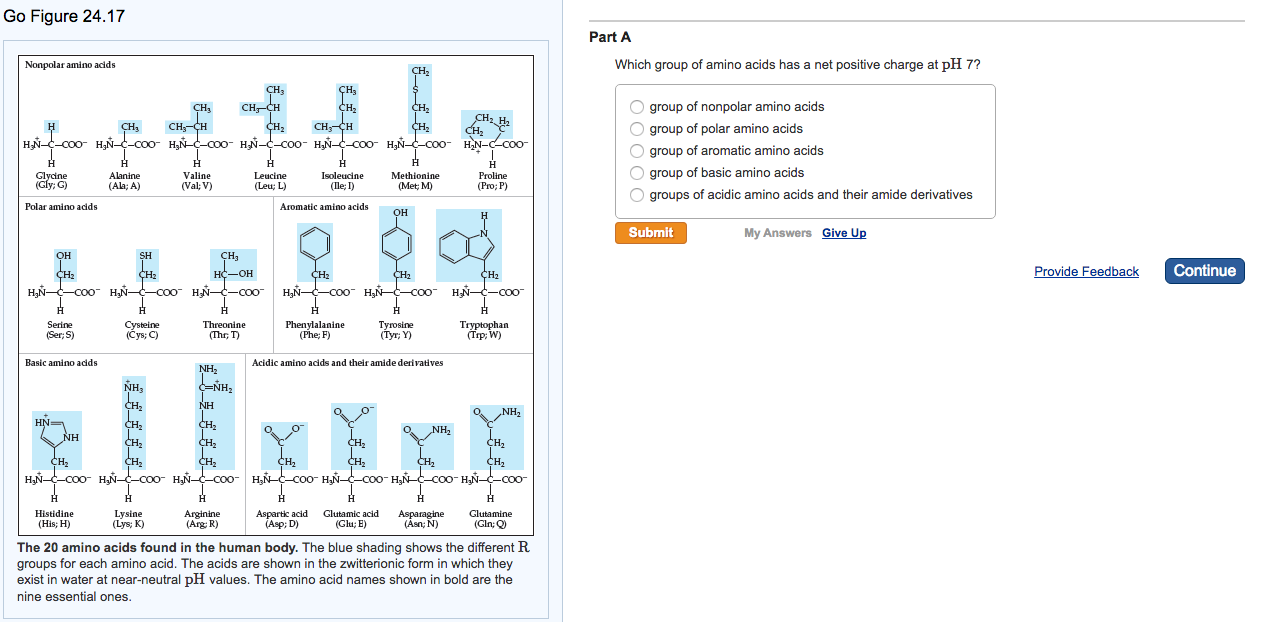
ALL *free* amino acids always have at least 2 chances to do so in their generic backbone, which has an amino group that can exist as -NH₃⁺ or -NH₂ & a carboxyl group which can exist as -COOH or -COO⁻. Other molecules are *weak* acids or bases they go back and forth more easily.Īmino acids are this weak kind. Some molecules, *strong* acids or bases have heavily weighted coins – they really want to give or take a proton and it’s hard to flip the coin back over to give it up again.
The logging also makes it so that small differences in the pH correspond to much more dramatic differences in H⁺ concentration. It’s a negative log of the *inverse* of the H⁺ concentration () which means that lower pH (more acidic) corresponds to higher H⁺ concentration and higher pH (more alkaline/basic corresponds to lower H⁺ concentration. Note: pH is a measure of proton availability. The pKa is the flipping power needed to flip half the coins. We can describe how “weighted” they are using pKa, which is the pH at which 1/2 the molecules are protonated, and the “flipping power” is the pH (lower pH (more acidic) means more protons available, helping flip the coin to the conjugate base side). So acid and base are 2 sides of the same coin (whose faces we call the conjugate acid & conjugate base), but the coin is “weighted” for different molecules so they prefer different forms. Giving away protons is *always* acting as an acid and once something acts as an acid it has to act as a base and take a H⁺ before it can give it up again. One definition of an acid is something that donates a proton (H⁺) and one definition of a base is something that accepts an H⁺. I would always get really confused about why we call the negatively charged amino acids “acidic” and the + charged ones “basic.” So let me try to explain. So, today let’s look at Arginine (Arg, R) And we can get a better appreciation and understanding of proteins if we look at those letters. Those generic parts are attached to a central “alpha carbon” (Ca), which is also attached to one of 20 unique side chains (“R groups”) which have different properties (big, small, hydrophilic (water-loving), hydrophobic (water-avoided), etc.) & proteins have different combos of them, so the proteins have different properties.
Positively charged amino acids free#
The reason for the “2 options” in parentheses is that these groups’ protonation state (how many protons (H⁺ ) they have) depends on the pH (which is a measure of how many free H⁺ are around to take).⠀ More on amino acids in general here but the basic overview is: amino acids have generic “amino” (NH₃⁺/NH₂) & “carboxyl” (COOH/COO⁻) groups that let them link up together through peptide bonds (N links to C, H₂O lost, and the remaining “residual” parts are called residues). Each day I’m going to bring you the story of one of these “charms” – what we know about it and how we know about it, where it comes from, where it goes, and outstanding questions nobody knows. There are 20 (common) genetically-specified ones, each with a generic backbone with to allow for linking up through peptide bonds to form chains (polypeptides) that fold up into functional proteins, as well as unique side chains (aka “R groups” that stick off like charms from a charm bracelet). Amino acids are the building blocks of proteins. It’s Day 15 of #20DaysOfAminoAcids – the bumbling biochemist’s version of an advent calendar.
